
Product Information
This page involves information on pharmaceutical products. However, it is not intended for advertising or providing medical advice.
Core Products
Recombinant human growth hormone product GROWJECT™
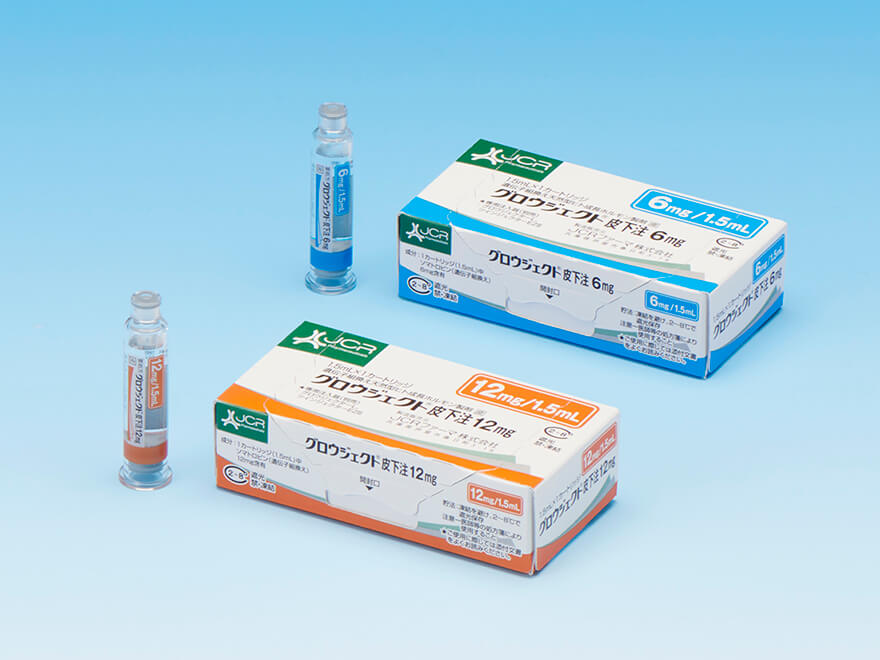
GROWJECT™ is a pharmaceutical indicated for the treatment of disorders such as pediatric short stature caused by the deficiency of growth hormone.
Launch
April 1993
Indication
- Growth hormone deficiency
- Turner syndrome
- Adult growth hormone deficiency
- Small for gestational age
- SHOX deficiency
Recombinant treatment for mucopolysaccharidosis type II IZCARGO™*1
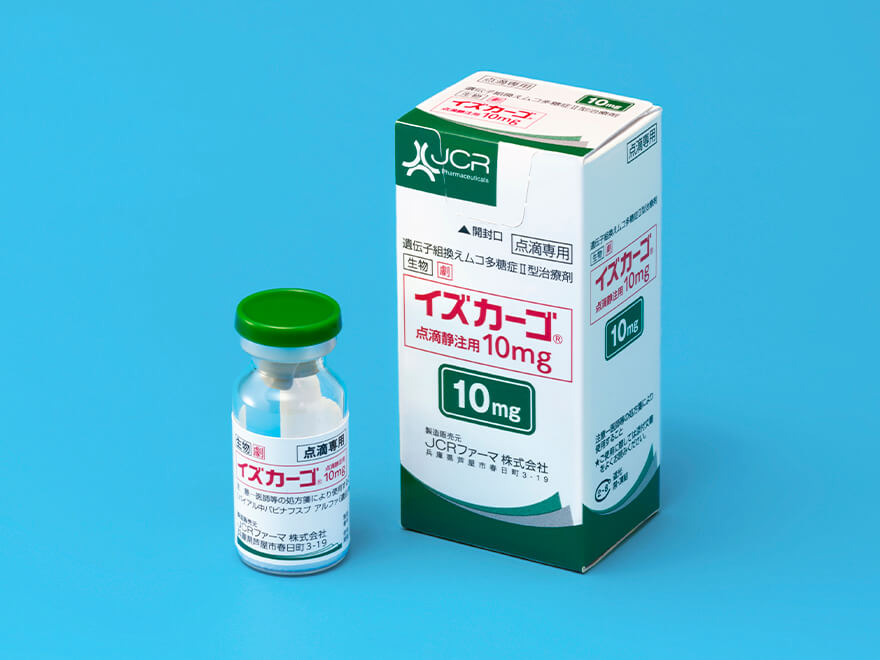
IZCARGO™ is the world’s first-ever approved enzyme replacement treatment (ERT) to apply JCR’s proprietary J-Brain Cargo® blood-brain barrier (BBB) penetration technology
Launch
May 2021
Indication
- Mucopolysaccharidosis type II
- *1Co-promotion with Sumitomo Pharma Co., Ltd from Apr.2023
Human somatic stem cell-processed products Human (allogeneic) bone marrow-derived mesenchymal stem cells TEMCELL™ HS Inj.

TEMCELL™ HS Inj. is Japan’s first allogeneic regenerative medical product manufactured by isolating and expanding mesenchymal stem cells derived from the bone marrow aspirate of a healthy adult donor, along with utilizing the function of the mesenchymal stem cells.
Launch
February 2016
Indication
- Acute GVHD following hematopoietic stem cell transplantation
Recombinant treatment for Fabry disease Agalsidase Beta BS I.V. Infusion [JCR]*2
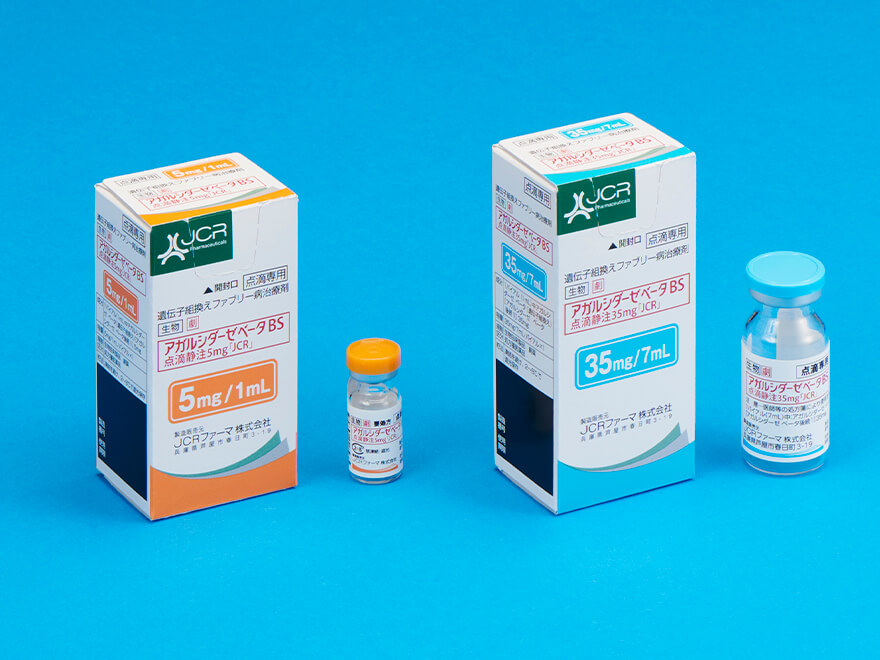
Agalsidase Beta BS I.V. Infusion [JCR] is JCR’s first enzyme replacement therapy (ERT) for lysosomal storage disorders (LSDs) and the first domestically produced ERT product for LSDs.
Launch
November 2018
Indication
- Fabry disease
- ※2JCR produces this product, and Sumitomo Pharma Co., Ltd. conducts pharmaceutical information dissemination activities and sales.
Recombinant erythropoietin product Epoetin Alfa BS Inj. [JCR]*3
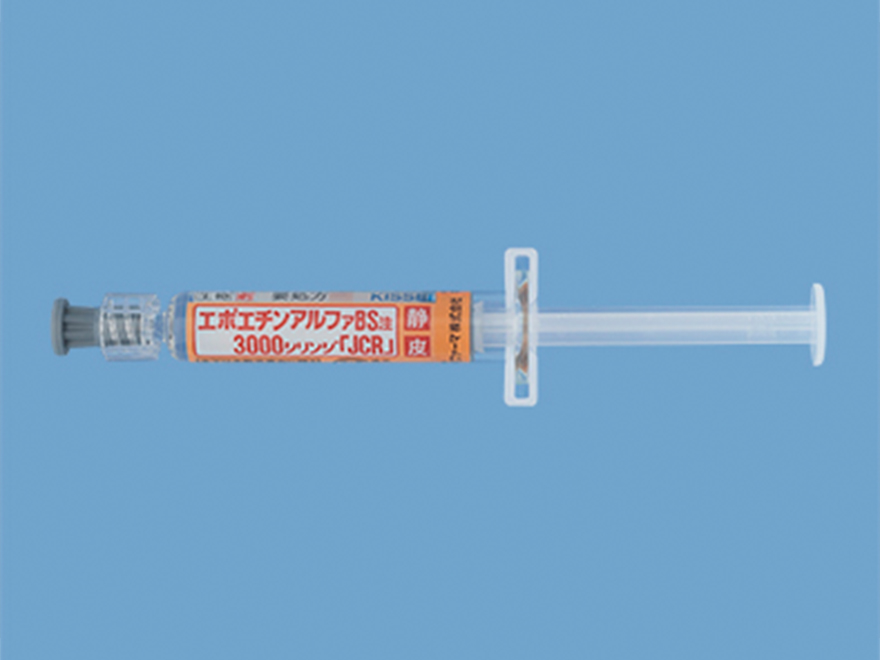
Epoetin Alfa BS Inj. [JCR] was developed utilizing our serum-free technology and proprietary biotechnologies. The product was launched as the first domestically produced biosimilar
Launch
May 2010
Indication
- Renal anemia in dialysis patient
- Anemia of prematurit
- ※3 JCR produces this product, and Kissei Pharmaceutical Co., Ltd. conducts pharmaceutical information dissemination activities and sales.
Long-acting erythropoiesis-stimulating agent Darbepoetin Alfa BS Inj. [JCR]*3
A biosimilar developed based on experience gained through Epoetin Alfa BS Inj. [JCR]
Launch
May 2021
Indication
- Renal anemi
- ※3JCR produces this product, and Kissei Pharmaceutical Co., Ltd. conducts pharmaceutical information dissemination activities and sales.